
Plasma Bile Acid Profiling Reveals Novel Early Diagnostic Biomarkers for Pediatric Sepsis
Introduction
Sepsis is a life-threatening condition that remains one of the leading causes of mortality and morbidity in critically ill children, particularly in pediatric intensive care units (PICUs).1 It is estimated that globally, 25.2 million children suffer from sepsis each year diagnosis and timely intervention are crucial in reducing the associated mortality.2 Pediatric sepsis is particularly difficult to diagnose early due to its atypical clinical manifestations and the non-specific nature of laboratory biomarkers. This leads to frequent misdiagnosis or delayed diagnosis, which significantly increases the risk of multi-organ failure and death. However, despite advances in diagnostic tools, there is currently no reliable, widely accepted diagnostic biomarker for early-stage pediatric sepsis.3 Currently, C-reactive protein (CRP) and procalcitonin (PCT) are the most studied biomarkers for severe sepsis, yet they exhibit significant limitations.4,5 While CRP is quickly measurable, its diagnostic accuracy is challenged by inter-individual variability and the potential for false-positive results, which can occur when CRP levels are elevated due to triggers such as inflammation from extravasation, cholestasis, or gastrointestinal pathology.6,7 Although studies suggest a reasonably good sensitivity for PCT in diagnosing sepsis (about 80%), its performance is not sufficient to serve as a standalone test. For example, in cases of suspected sepsis, one in five patients may go undiagnosed if solely relying on PCT.8
In recent years, metabolomics has emerged as a promising approach in the study of pediatric sepsis. Several studies have employed metabolomics to identify differentially expressed metabolites between sepsis patients and healthy controls, uncovering significant alterations in metabolic pathways, including amino acid biosynthesis, phenylalanine metabolism, tyrosine metabolism, and glycine, serine, and threonine metabolism, among others.9-11 Additionally, Mickiewicz et al integrated metabolomics with inflammatory mediators to differentiate between children who required intensive care and those who did not, providing valuable insights for sepsis phenotyping and risk stratification.12 However, while metabolomics has identified sepsis-related metabolic disruptions in adults, pediatric-specific bile acid alterations remain underexplored.
Given that sepsis is characterized by a dysregulated host response to infection, certain metabolic pathways may serve as critical indicators of disease progression and severity. Among these pathways, bile acid (BA) metabolism has garnered increasing attention due to its multifaceted role in gut metabolism, cell signaling and immune regulation. Bile acids, including primary bile acids (PBAs) and secondary bile acids (SBAs), are not only involved in lipid digestion but also act as signaling molecules that regulate inflammation and cellular stress responses.13 Accumulating evidence suggests that bile acid dysregulation occurs in inflammatory diseases. For instance, in patients with inflammatory bowel disease (IBD), significant reductions in serum secondary bile acids have been observed.14 Bile acids regulate intestinal barrier function and immune responses by activating nuclear receptors (FXR, PXR) and membrane receptors (TGR5).15 Secondary bile acids, including lithocholic acid (LCA) and deoxycholic acid (DCA), can inhibit macrophage inflammatory responses via the TGR5 signaling pathway.16 These insights suggest that bile acids may play a pivotal role in sepsis-induced immune-metabolic dysfunction. However, their potential as early diagnostic biomarkers for pediatric sepsis remains largely unexplored. In this study, we conducted a two-phase research design, comprising an untargeted metabolomics screening followed by targeted bile acid quantification for validation. The aim was to identify potential metabolic predictors of pediatric sepsis.
Materials and Methods
Patients and Sample Collection
The study cohort consisted of pediatric sepsis patients admitted to two tertiary-care PICUs in Beijing between 2015 and 2017. Sepsis diagnosis in these patients was based on the China Pediatric Sepsis Consensus (2015) and the Surviving Sepsis Campaign International Guidelines (2012).17,18 The inclusion and exclusion criteria for pediatric sepsis patients have been previously described in our earlier studies.19-21 Inclusion criteria: (1) community-acquired sepsis; (2) Children aged 1 month to 12 years; (3) admission to the PICU within 24 hours after sepsis diagnosis; (4) laboratory results obtained within 24 hours of PICU arrival. Exclusion criteria: (1) patients with diseases that affect metabolism, such as food or drug poisoning, cholestasis, biliary atresia, chronic liver disease, chronic diarrhea, immunodeficiency, malnutrition, congenital metabolic diseases, and other related conditions; (2) severe congenital malformations or trauma; (3) Use of drugs affecting metabolism within one month prior to admission, such as: Steroids, immunosuppressants, and intravenous nutrition; (4) Cases with significant missing data due to refusal of laboratory tests or other reasons after admission. The healthy control group consisted of children with no physical discomfort and normal results in routine blood tests, including blood count, blood biochemistry, and other relevant tests.
Sepsis patients were enrolled within 24 hours of admission, and fasting blood samples were collected from the peripheral vein using EDTA-coated tubes (0.5-1 mL) to obtain plasma for subsequent analysis. Similarly, healthy pediatric controls were selected from children undergoing routine health check-ups during the same period. These controls were matched based on age and sex, and blood samples were also collected in a fasting state. After collection, all samples were labeled, processed, and centrifuged. The resulting plasma was transferred into EP tubes and stored at −80 °C in a ultra-low temperature freezer for long-term storage until further analysis. Clinical data, including patient demographics (name, sex, age), as well as relevant clinical information, were also recorded for all enrolled subjects.
This study was approved by the Ethics Committee of the Capital Medical University Affiliated Children’s Medical Center (formerly named the Capital Institute of Pediatrics; approval number SHERLL 2013082), and written informed consent was obtained from the legal guardians of all participants. This study was conducted in accordance with the Declaration of Helsinki.
Non-Targeted UPLC-MS Metabolomics Analyses
Serum Samples stored at −80 °C were thawed on ice until no ice crystals remained. An 80 µL aliquot of each sample was mixed with 10 µL of internal standard L-2-chlorophenylalanine (0.3 mg/mL, prepared in methanol) and vortexed for 10 seconds. Subsequently, 240 µL of protein precipitant (methanol: acetonitrile, 2:1 v/v) was added, followed by vortexing for 1 minute. The mixture was then submitted to ultrasonic extraction with an ice water bath for 10 min and kept at −20°C for 30 minutes. After centrifugation at 13,000 rpm for 15 minutes (4 °C), 150 µL of the supernatant was aspirated using a syringe, filtered through a 0.22 µm organic phase filter, and transferred to an LC injection vial for storage at −80 °C until LC-MS analysis. Quality control (QC) samples were prepared by pooling equal volumes of extracts from all samples, with each QC sample volume matching that of the individual samples.
The analytical platform consisted of a Waters ACQUITY UPLC (Waters, Milford, USA) system coupled with an AB Sciex Triple TOF 5600 high-resolution mass spectrometer (AB SCIEX, USA). The liquid chromatography conditions included an ACQUITY UPLC BEH C18 column (100 mm × 2.1 mm, 1.7 µm), maintained at 45°C. The mobile phase consisted of solvent A (water with 0.1% formic acid) and solvent B (acetonitrile with 0.1% formic acid), with a flow rate of 0.4 mL/min and an injection volume of 5 µL. The gradient program is detailed in Table S1. Mass spectrometry data were acquired using electrospray ionization (ESI) in both positive and negative ion modes, with specific parameters provided in Table S2.
Figures S1A and S1B display the base peak ion chromatograms (BPC) of QC samples in positive and negative ion modes, respectively. Visual inspection of BPCs for all samples confirmed strong signal intensity, high peak capacity, and excellent retention time reproducibility. Raw data were processed using the metabolomics software Progenesis QI for baseline filtering, peak identification, integration, retention time correction, peak alignment, and normalization, generating a data matrix of retention time, mass-to-charge ratio (m/z), and peak intensity.
Targeted Quantitative Analysis of Bile Acid
A 20 µL aliquot of plasma was transferred to a 1.5 mL Axygen tube and mixed with 80 µL of deuterium-labeled bile acid internal standards (0.1 µM d4-CA, d4-CDCA, d4-DCA, d4-LCA; 10 µM d4-TDCA, d4-TCA, d4-GUDCA, d4-GCA, d4-TLCA, d4-GLCA in methanol). The mixture was vortexed for 1 minute and centrifuged at 120,00 rpm for 20 minutes (4°C). The supernatant was transferred to an injection vial for analysis. A standard curve was generated by processing 20 µL of standard solutions (0-100 µM) using the same protocol. A standard curve was deemed acceptable when the coefficient of determination (R2) reached 0.99.
The analysis was performed using a Shimadzu LC-20AD pump system coupled with an API 6500 Q-TRAP mass spectrometer (AB SCIEX, Framingham, MA). The supernatant (70 µL) was injected onto a BEH C18 UPLC column (2.1 × 150 mm, 1.7 µm) at a flow rate of 0.35 mL/min. A gradient elution was employed, starting with 25% B and increasing to 40% B over 12 minutes, followed by a rise to 75% B over 14 minutes. The column was washed with 100% B for 2 minutes and equilibrated with 25% B for 4 minutes between injections. The column temperature was maintained at 45°C. Analytes were detected using negative ion mode electrospray ionization, with multiple reaction monitoring (MRM) of precursor and characteristic product ion transitions. Concentrations of the following bile acids were quantified: cholic acid (CA), DCA, chenodeoxycholic acid (CDCA), ursodeoxycholic acid (UDCA), LCA, glycocholic acid (GCA), glycodeoxycholic acid (GDCA), glycochenodeoxycholic acid (GCDCA), glycoursodeoxycholic acid (GUDCA), taurocholic acid (TCA), taurodeoxycholic acid (TDCA), and taurochenodeoxycholic acid (TCDCA).
Statistical Analysis
Normally distributed continuous data were expressed as mean ± standard deviation and compared using the independent samples t-test. Non-normally distributed continuous data were expressed as median (interquartile range) [M (P25, P75)] and analyzed using the Wilcoxon rank-sum test. Categorical data were summarized as counts (percentages), and between-group differences were assessed using the chi-square test.
For non-targeted metabolomics, unsupervised principal component analysis (PCA) was performed using the prcomp function in R to explore metabolic profile differences between groups. Differential metabolites were identified through univariate and multivariate analyses. Univariate analysis included fold change (FC) calculations and t-tests. Multivariate orthogonal partial least squares-discriminant analysis (OPLS-DA) was conducted using the MetaboAnalystR package to establish a classification model, with variable importance in projection (VIP) scores calculated for all detected metabolites. Metabolites meeting both VIP > 1 and p < 0.01 were considered statistically significant. Pathway enrichment and metabolic pathway analyses of differential metabolites were performed using the Kyoto Encyclopedia of Genes and Genomes (KEGG) database.
For targeted bile acid quantification, concentrations of bile acids were compared between groups using the t-test. Binary logistic regression analysis was employed to evaluate the association between differential bile acid levels and sepsis risk. To identify robust and reproducible bile acid biomarkers, five machine learning algorithms with distinct feature selection strategies were applied to the bile acids that reached significance in univariate analysis. These included: (1) Recursive Feature Elimination (RFE), a wrapper-based technique that recursively eliminates the least informative features based on model performance metrics; (2) Least Absolute Shrinkage and Selection Operator (LASSO), a regularized regression method that introduces an L1 penalty to perform variable selection by shrinking non-contributory coefficients to zero; (3) Random Forest (RF), a tree-based ensemble learning method that ranks variable importance by assessing each feature’s contribution to impurity reduction across multiple decision trees; (4) Support Vector Machine-Recursive Feature Elimination (SVM-RFE), which utilizes the weight vectors of a linear support vector machine to guide iterative elimination of less relevant features; and (5) Gradient Boosting Decision Tree (GBDT), a boosting technique that constructs an additive model of decision trees in a forward, stage-wise manner to optimize predictive accuracy while generating feature importance scores. Receiver operating characteristic (ROC) curve analysis was conducted to assess the diagnostic performance of bile acids. Odds ratios (ORs) with 95% confidence intervals (CIs) and area under the curve (AUC) values were reported to demonstrate statistical and diagnostic significance.
Results
Demographic Characteristics of the Included Population
A total of 100 participants were enrolled in the discovery phase, including 50 children with sepsis and 50 age- and sex-matched healthy controls. In the validation phase, 141 participants were recruited, comprising 61 sepsis patients and 80 matched healthy individuals. Baseline characteristics of the two cohorts are presented in Table 1. Significant differences were observed between groups in white blood cell (WBC) count, CRP, PCT, alanine aminotransferase (ALT), and aspartate aminotransferase (AST). Total bile acid (TBA) and total bilirubin (TBIL) levels remained within the normal range and showed no statistically significant differences between groups.
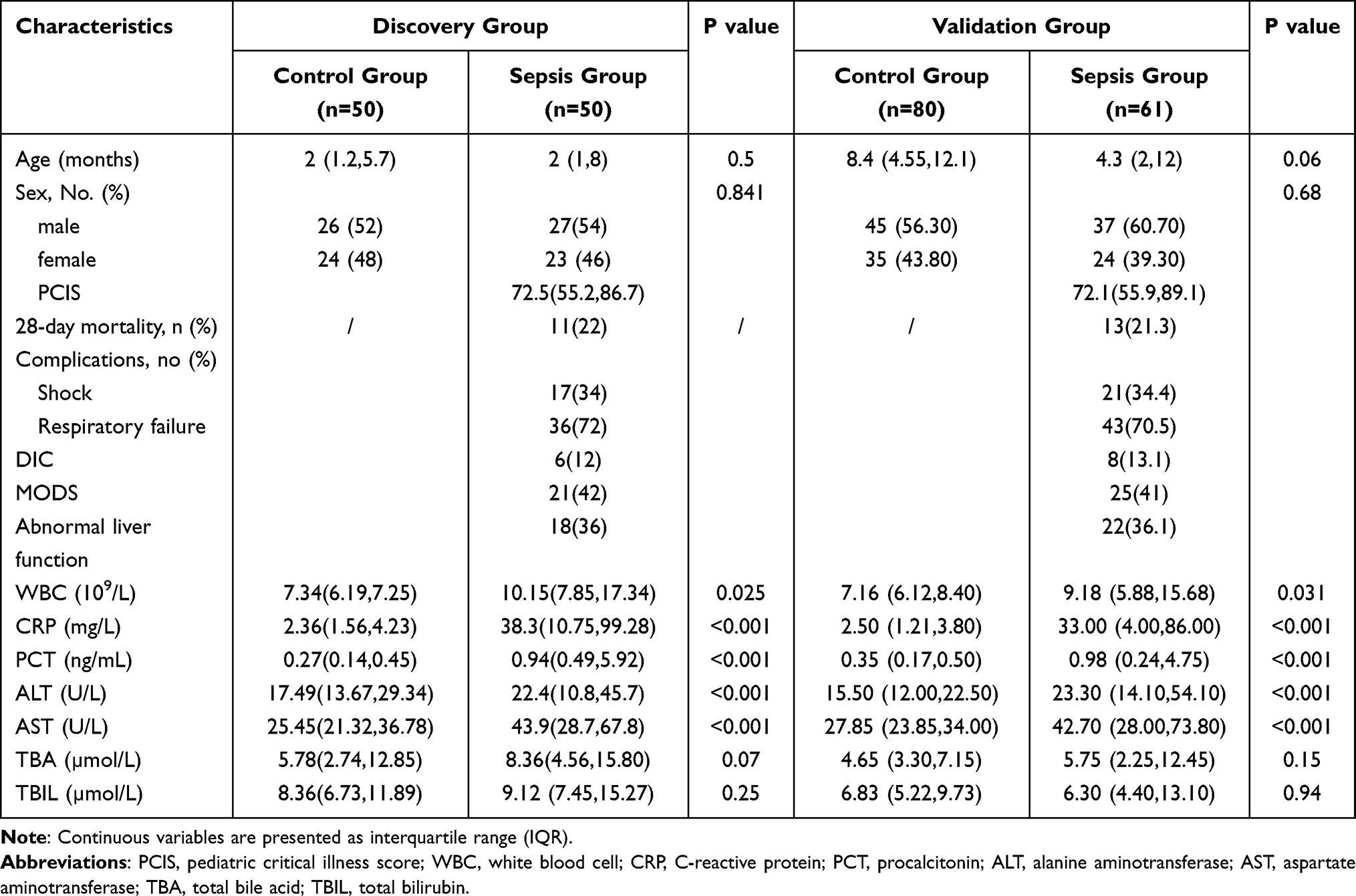
Table 1 Clinical Characteristics of Patients Enrolled in This Study
Non-Targeted UPLC-MS Metabolomics Analyses
In the discovery cohort, PCA score plots showed clear separation between sepsis and control groups. Partial least squares discriminant analysis (PLS-DA) and OPLS-DA confirmed this separation with improved clustering and discrimination performance (Figure 1). A total of 347 significantly altered metabolites were identified, of which 134 were upregulated and 213 were downregulated. These included sphingolipids, glycerophospholipids, arachidonic acid metabolites, bile acids, steroid hormones, and amino acid derivatives (Table S3). KEGG enrichment analysis of these metabolites revealed disruptions in primary bile acid biosynthesis, steroid hormone metabolism, and pathways involving unsaturated fatty acids and arachidonic acid (Figure 2).
Figure 1 Multivariate statistical score plots of the sepsis screening cohort. (A) Principal component analysis (PCA) plot; (B) Partial least squares discriminant analysis (PLS-DA) plot; (C) Orthogonal partial least squares discriminant analysis (OPLS-DA) plot.

Figure 2 KEGG enrichment analysis of differential metabolites in non-targeted metabolomics.
Targeted Quantitative Analysis of Bile Acid
Targeted quantification of bile acid concentrations was performed in the validation cohort. Table 2 summarizes the levels of 12 bile acids in the two groups. Significant differences were observed in the concentrations of LCA, DCA, UDCA, CA, GUDCA, GCDCA, GCA, TDCA, TCA, GDCA and TCDCA. Compared with controls, patients with sepsis showed elevated levels of primary bile acids (CA, GCA, TCA, GCDCA, TCDCA) and conjugated secondary bile acids (TDCA), while levels of secondary bile acids (LCA, DCA, UDCA) and their glycine conjugates (GUDCA, GDCA) were significantly decreased. Moreover, the CA/CDCA ratio, representing the balance between the classic and alternative pathways of primary bile acid synthesis, was significantly increased (p < 0.001). Indicators of secondary bile acid formation such as DCA/CA, GDCA/CA, and LCA/CDCA were decreased. The TDCA/DCA ratio was elevated, while the GDCA/DCA ratio was reduced (p < 0.05) (Table 2).
Table 2 Levels of Serum Bile Acids in the Control Group and the Sepsis Group During the Validation Stage
Machine Learning Identifies Key Bile Acid for Sepsis Diagnosis
Univariate binary logistic regression was applied to bile acid and clinical variables (Table 3). WBC, CRP, and PCT were significantly associated with sepsis, with PCT showing the highest odds ratio (OR = 8.42, 95% CI: 2.51-28.28). Bile acids including TCA, TDCA, GCA, TCDCA, GCDCA, and TDCA/DCA were positively associated with sepsis risk, with ORs of 39.06, 29.39, 4.59, 2.63, 1.56, and 6.08 respectively. Conversely, LCA, DCA, UDCA, GUDCA, and DCA/CA had ORs < 1, indicating protective associations.

Table 3 Univariate Binary Logistic Regression Analysis of Clinical and Bile Acid Indicators
Five machine learning algorithms (RFE, LASSO, RF, SVM-RFE, and GBDT) were used to further evaluate the 11 bile acids identified as significant predictors. RFE selected 8 bile acids (Figure 3A). The optimal LASSO model included 11 variables with top coefficients for LCA, TDCA, and GUDCA (Figures 3B and C). RF showed the highest accuracy and kappa when using 7 features (Figure 3D). SVM-RFE identified 9 bile acids as optimal predictors (Figure 3E), while GBDT selected 11 features, with GCA, UDCA, and GUDCA ranked as the top 3 (Figure 3F). Feature overlap among models is shown in Figure 3G and detailed in Table S3. The top four bile acids common across models were UDCA, GUDCA, GCA, and DCA. Their individual predictive performance, assessed via ROC curves (Figure 3H, Tables 4 and S4), yielded AUCs of 0.775, 0.767, 0.757, and 0.748, respectively. Combined, these four markers achieved an AUC of 0.880, indicating excellent predictive accuracy.
Table 4 Accuracy of Logistic Regression Model in Diagnosing Sepsis

Figure 3 Machine learning identification of 11 intersect bile acid features during the validation stage. (A) Performance evaluation of Recursive Feature Elimination (RFE) algorithm for feature selection; (B) Coefficient path plot from LASSO regression, with two dashed lines indicating the minimum λ (lambda.min) and the maximum λ within one standard error (lambda.1se), suggesting that selecting λ between these two values is reasonable; (C) Selected features and their corresponding coefficients from LASSO; (D) Accuracy and Kappa values of the Random Forest (RF) mo (E) Relationship between the number of variables and model accuracy in the Support Vector Machine Recursive Feature Elimination (SVM-RFE) algorithm; (F) Variable importance scores in the Gradient Boosting Decision Tree (GBDT) model; (G) Upset plot showing the intersection of features selected by five machine learning algorithms: the top bar chart displays the number of bile acid species unique to or shared among algorithms, the left bar chart indicates the total number of variables selected by each algorithm, and the central matrix uses single-color nodes to represent variables unique to a specific algorithm and connected nodes to represent variables shared among multiple algorithms; (H) Receiver Operating Characteristic (ROC) curves for variables selected by the five algorithms.
Predictive Value of Bile Acids for Sepsis-Related Complications
In the validation cohort comprising 61 pediatric sepsis patients, subgroup analyses were conducted based on the presence of septic shock and respiratory failure (definitions provided in Box S1). Patients were stratified into shock and non-shock subgroups. Comparative analysis of bile acid levels between these groups revealed statistically significant differences in the ratios of deoxycholic acid to cholic acid (DCA/CA), GCDCA, and CDCA (Table S5). Similarly, patients were categorized into respiratory failure and non-respiratory failure subgroups. Significant differences were observed in LCA, CA, and LCA/CDCA between these groups (Table S6).
Univariate logistic regression analyses were performed for the aforementioned bile acid indicators (Figure 4). The results indicated that each unit increase in the DCA/CA ratio was associated with a 4.5% increase in the odds of developing septic shock (OR = 1.045, 95% CI: 1.009-1.081). Conversely, each unit decrease in GCDCA was linked to a 30% decrease in the odds of septic shock (OR = 0.700, 95% CI: 0.526-0.932). Furthermore, elevated levels of LCA were identified as a risk factor for respiratory failure in sepsis patients (OR = 2.154, 95% CI: 1.022-4.540).
Figure 4 Forest plot of univariate logistic regression analysis of bile acids associated with septic shock or respiratory failure.
Discussion
This study utilized untargeted metabolomics to identify dysregulation in the primary bile acid (BA) biosynthesis pathway in pediatric sepsis. Subsequent targeted quantification analyses confirmed significant alterations in the BA profile of septic children. Specifically, elevated levels of primary BAs—CA, GCA, TCA, GCDCA, and TCDCA—and the taurine-conjugated secondary BA TDCA were observed. Conversely, secondary BAs such as DCA, GDCA, UDCA, GUDCA, and LCA were significantly reduced. These findings suggest that while total bile acid (TBA) levels remain stable during the early stages of sepsis, there are significant changes in the composition of individual bile acids, which may serve as potential biomarkers for diagnosing and predicting sepsis and its complications.
The “gut-liver axis” is recognized as a critical component in the pathogenesis of sepsis. Experimental data support the notion that the gut and liver influence each other through mechanisms such as bacterial translocation and the modulation of the microbiome by liver-derived molecules like Bas.22 BAs can also regulate metabolism and inflammation through interactions with various receptors. Consequently, BA metabolism is increasingly acknowledged as a significant factor in the early prediction and diagnosis of diseases. In adult sepsis patients, metabolomic analyses have revealed decreased levels of short-chain fatty acids and secondary BAs, alongside increased levels of primary BAs.23 However, studies focusing on the association between BAs and pediatric sepsis are limited. A report indicated that serum BA tauro-omega-muricholic acid (TOMCA) is an independent factor associated with early-onset neonatal sepsis.24 Additionally, animal studies have demonstrated that intravenous administration of TDCA can reduce serum pro-inflammatory cytokines, normalize hypotension, prevent renal injury, and prolong survival in septic mice.25
In our study, there was no statistically significant difference in TBA levels between the sepsis and control groups during the early stages of the disease, with levels remaining within the normal range. However, the concentrations of primary BAs (CA, GCA, TCA, GCDCA, TCDCA) and secondary BAs (TDCA) were significantly elevated, while the secondary BAs (DCA, GDCA, UDCA, GUDCA, LCA) showed a marked decrease. These findings suggest that although total bile acids remain unchanged in the early stages of sepsis, there is already a significant alteration in the composition of individual BAs in pediatric sepsis patients. As the disease progresses, previous studies have shown that sepsis patients who died during hospitalization had significantly higher and more fluctuating levels of bile acids than survivors, with statistically significant differences.26
We observed an increased CA/CDCA ratio in the sepsis group compared to controls, suggesting disruption of the classical and alternative pathways of primary BA synthesis during sepsis. Under physiological conditions, the classical pathway produces over 90% of total bile acids, while the alternative pathway accounts for less than 10%, making the classical pathway the dominant biosynthesis route.27 The increased CA/CDCA ratio in our study indicates enhanced activity of the classical pathway in early pediatric sepsis. CYP8B1, a key enzyme in the BA synthesis pathway, determines the CA/CDCA ratio by regulating the production of CA in the classical pathway.28,29 We hypothesize that CYP8B1 may be involved in the altered BA metabolism observed in early sepsis.
Primary BAs are further metabolized by the gut microbiota into secondary BAs.29,30 The decrease in the LCA/CDCA, DCA/CA, and GDCA/CA ratios suggests a reduction in secondary BA production due to microbiota dysbiosis, as primary BAs undergo hydrolysis and dehydrogenation by the gut microbiota. Additionally, the lowered GDCA/DCA ratio and elevated TDCA/DCA ratio in the sepsis group indicate a shift from glycine-conjugated to taurine-conjugated secondary BAs, suggesting an altered microbial metabolism. Similar findings were reported in full-term neonates with early-onset sepsis, where glycine-conjugated BAs were significantly lower compared to controls.31
The odds ratios (ORs) of GCA, GCDCA, TCA, TCDCA, TDCA, and TDCA/DCA were greater than 1, indicating that higher concentrations of these BAs in the early stages of sepsis are associated with an increased risk of sepsis. Conversely, the ORs for LCA, DCA, UDCA, GUDCA, and DCA/CA were less than 1, suggesting that lower concentrations of these BAs are associated with an increased likelihood of developing sepsis. These findings underscore the potential role of primary conjugated BAs and taurine-conjugated secondary BAs in increasing sepsis risk, while decreased secondary BA levels may also contribute to the development of sepsis. BAs and gut microbiota share a bidirectional relationship: BAs can shape the gut microbiota, while the microbiota can alter the BA pool.32 Dysbiosis of the gut microbiota has been identified as a risk factor for sepsis and subsequent organ dysfunction.33 Thus, strategies to manipulate the gut microbiota, such as fecal microbiota transplantation, could potentially benefit sepsis patients by restoring microbial balance.
Our machine learning models identified a combination of UDCA, GUDCA, GCA, and DCA as key diagnostic indicators for sepsis. UDCA, a secondary BA converted from chenodeoxycholic acid by intestinal bacteria, has been shown to have immunomodulatory and anti-apoptotic properties, as well as the ability to prevent hepatocyte cytokine release. Animal studies have demonstrated that UDCA primarily exerts its protective effects through activation of the Nrf2/HO-1 pathway and inhibition of the NF-κB pathway, reducing oxidative stress and inflammation, and protecting against sepsis-induced acute kidney injury (AKI).34 UDCA also blocks PANoptosis via the STING pathway to mitigate sepsis-induced lung injury.35 Oral administration of UDCA in neonatal mice showed significant antimicrobial effects and helped maintain colonic barrier integrity in peritonitis and sepsis models.36 Despite the fact that the use of UDCA in critically ill adults with sepsis/septic shock has not shown improvement in shock severity or resolution, patients treated with UDCA were more likely to be extubated by day 3 in the ICU and did not require mechanical ventilation.37 GUDCA has been shown to reduce the production of lactate dehydrogenase, TNF-α, and IL-1β in neurological models, as well as decrease the production of cytochrome c peroxidase in neurodegenerative disease models.38 Although research on UDCA and GUDCA in sepsis is limited, its promising effects suggest that it could potentially serve as an alternative therapeutic approach for such diseases.
Furthermore, our study identified GCDCA and the DCA/CA ratio as important predictive biomarkers for septic shock, while LCA was found to be a predictor of sepsis-related respiratory failure. In vitro experiments have demonstrated that GCDCA induces liver fibrosis through the NLRP3 inflammasome pathway.39 However, the mechanisms underlying the reduction of GCDCA and its association with an increased risk of septic shock warrant further investigation. Secondary bile acids, such as LCA and DCA, have been reported to mitigate intestinal inflammation. This is consistent with studies suggesting that bile acids play a crucial role in regulating pulmonary function and inflammation. For instance, a study by Yu et al showed that an increase in maternal bile acids impaired fetal lung development and pulmonary surfactant production, leading to respiratory failure and poor perinatal outcomes.40 Similar findings have been observed in lung transplant recipients, where TCA (taurocholic acid) and GCA (glycocholic acid) levels in large airway bronchial wash (LABW) were correlated with acute lung allograft dysfunction and death. Elevated TCA and GCA in LABW were associated with worsened lung function post-transplant, and higher levels of TCA were predictive of death in these patients.41 This highlights the complex interplay between bile acids and microbial metabolism in the pathophysiology of sepsis and emphasizes the need for further research to explore how bile acids influence both pulmonary and intestinal function in septic patients.
Our study also suggests potential therapeutic implications, particularly regarding secondary bile acids with immunomodulatory and hepatoprotective properties. Previous studies have shown that ursodeoxycholic acid (UDCA) and its derivative, obeticholic acid, can improve bile acid homeostasis, inhibit the expression of TNF-α, IL-6, and IL-1β, and alleviate sepsis-associated liver injury.42 These findings indicate that bile acids may have a protective role in sepsis. Additionally, a study by Ainosah et al demonstrated that UDCA was beneficial in treating and preventing sepsis-induced cholestasis in experimental animals.43 This effect was observed through improvements in liver enzyme expression levels and tissue histopathological abnormalities, further supporting the protective role of bile acids in sepsis.
There are several limitations to our study. Our study did not include neonates in the validation cohort, and future research should address this gap to determine whether the observed alterations in BA profiles are consistent across different age groups. The cross-sectional design of the study prevents us from establishing causal relationships between BA alterations and the progression of sepsis. Diet and antibiotics administered during hospitalization may influence bile acid metabolism by affecting gut microbiota. Although we excluded prior antibiotic use, the potential impact of antibiotics during the ICU stay on bile acid profiles should be considered in future studies.
Conclusions
This study demonstrates that bile acid metabolism is significantly altered in pediatric patients with sepsis during the early disease stage. Specifically, an imbalance between primary and secondary BAs, as well as between glycine- and taurine-conjugated forms, was observed. Machine learning algorithms identified four key bile acids-UDCA, GUDCA, GCA, and DCA-as potential diagnostic biomarkers for pediatric sepsis, with high predictive accuracy. Furthermore, specific BAs such as GCDCA and the DCA/CA ratio were associated with the risk of septic shock, while LCA was linked to respiratory failure, suggesting that BAs may also serve as prognostic markers for sepsis-related complications. Taken together, our findings provide novel insights into the role of BAs in pediatric sepsis. While these bile acids show potential as early diagnostic and prognostic biomarkers, future studies should validate these biomarkers in neonates and assess their dynamic changes during sepsis progression. Additionally, multicenter studies involving demographically diverse populations are necessary to validate the clinical utility of these biomarkers, considering geographic and age-related variability in the disease.
Link nội dung: https://melodious.edu.vn/anh-sex-vip-a98156.html